Atomic absorption spectroscopy AAS and atomic emission spectroscopy AES is a spectroanalytical procedure for the quantitative determination of chemical elements using the absorption of optical radiation light by free atoms in the gaseous stateAtomic absorption spectroscopy is based on absorption of light by free metallic ions. Follow steps 3- 7 for the 380 nm wavelength.

Chromatographic Calculations

The Integrated Absorbance Of The Peak Area Between 1390 And 1250 Cm 1 Download Scientific Diagram

Illustrations Of Peak Area And Peak Height Measurements A Peak Area Download Scientific Diagram
Close the door and read the Absorbance shown.

Is it peak area is the same as absorbance. Measured as peak area or peak height is. The blood serum level ofCarbamazepine is estimated as 145 ppm. Standards are measured first and can consist of either one or.
Peak Area against Serum Carbamazepine Level 0. If aqueous solutions are measured the microplate measurement can be normalized to 1 cm by using the water-peak path length correction. Beers law states that the sample absorbance is directly proportional to concentration.
Press read Set cursor mode to peak and valley under Options More Cursor Mode Press the left and right arrows to the right of the graph to select the highest peak. From the equation for the best-fit line for the absorbance at 525 nm as a function of Al 3 determine the percent aluminum by weight in alum and share your results with the class. The method of standard addition is a type of quantitative analysis approach often used in analytical chemistry whereby the standard is added directly to the aliquots of analyzed sample.
In analytical chemistry the technique is used for. Integrated absorbance not peak absorbance depends linearly on concentration. Area measurements are more linear than peak height measurements because most of the area of a peak is measured when the transient absorbance is less than maximum and where Beers Law is more strictly obeyed.
The peak area can be correlated to concentration using a simple calibration curve of a range of standard solutions like in this example of popular soda ingredients. For instance the absorbance of the alkene 2-methyl pent-2-ene is below 200 nm as is the ππ absorbance of 4-methyl pentane-2-one below. In addition to overlaying signals their ratios ca be calculated and plotted.
Perform a Q-test on the class data and then calculate the average percent Al by weight in alum the standard deviation of the data and. The SpectraMax ABS and ABS Plus absorbance microplate readers provide the flexibility sensitivity and convenience for a wide range of assays such as ELISAs microbial growth and protein quantitationBoth readers feature an 8-channel read head for fast reads and a subset feature the temperature independent. The system which produced signal C provided twice the signal or sensitivity of that which produced signal D but also generated twice the noise level.
Determine optimum wavelength absorbance and set up data collection mode. Calculate the concentration of each component in the diet sodas from the peak areas. The optical absorbance spectra contains information on nanosphere or nanorod quality.
As a result perfor-mance should also be the same for the two systems. Avobenzone C20H22O3 CID 51040 - structure chemical names physical and chemical properties classification patents literature biological activities safety. Each sample application contains approximately the same quantity by weight of material to be chromatographed.
3- Both sample absorption peak complex and the ligand absorption peak blank are overlaps at the same region no sufficient peaks resolutionand that also meanyou may have higher ligand conc. So if the peak absorbance is below 10 then using. The most probable transition causes the most intense peak.
The amount of energy required for the transition depends on the nature of the two orbitals involved in the bonding and that is why a simple functional group always causes absorption in the same general area. Place a cuvette with a sample into the spectrophotometer. Thanks to its high-speed built-in CCD detector the Duetta can acquire a full spectrum from 250 nm to 1100 nm in less than one second making it the fastest fluorescence spectrometer on the market.
Using the peak area and known concentrations create a calibration curve for each component and determine the least-squares fit for each calibration curve. We provide the peak width at 80 of the maximum peak as a measure of this quality on our tech spec sheets. Calculate the peak height times with the width at half of the height and use this value as the peak area.
The narrower the peak width the smaller the standard deviation of particle diameter or aspect ratio respectively. On the other hand peak height measurements are simpler to make and are less prone to interference by neighboring overlapping peaks. If the dominant source of noise is photon noise the precision of absorbance measurement is theoretically best when the absorbance is near 10.
Therefore absorbance acquired in microplates is typically lower than the absorbance of the same solution measured in cuvettes. In some cases where the solution matrix interferes with the measurement of the solute a classical calibration curve can be inaccurate. Where a is the spectral absorbance A is the IA ie the area under the curve I 0 ν and I t ν are the incident light intensity and the transmitted light intensity respectively P X m is the partial pressure of absorber m S m T is the line strength L is the absorption length and Φ ν is a line-shape function whose integral is normalized to unity Φ ν d.
In mesityl oxide where the alkene and CO group are in conjugation with each other the absorption maximum moves to longer wavelength at 228 nm. The signal on the left seems to be a more-or-less straight line but its numerically calculated derivative dxdy plotted on the right shows that the line actually has several approximately straight-line segments with distinctly different slopes and with well-defined breaks between each segment. The numerical integration of the absorbance leads to maximum deviations from linearity of less than 01.
Data can be collected as either peak height or area. Absorbance or to equal areas. Filterless readers provide consistent results with steady temperature regulation.
Continue this same process to complete Data Table 2. Quantitative measurements are possible by means of densitometry absorbance or fluorescence measurements. 2 Quant Mode - Quantization is usually performed on a major peak of the sample.
Record the wavelength and absorbance in. The method is shown graphically for a blood serum sample of peak area293 mm 2. Again are for the same concentration.
Nominal volumes of the volumetric glassware used in this exercise. The signal-to-noise ratios for the two signals are therefore the same SN 8 for each. Thus number of wavelengths are absorbed in the same vicinity.
The resulting ratiograms are sensitive indicators of peak purity. Peak Area mm 2 Using this calibration line plot and trendline the unknown concentration of Carbamazepine in a serum sample may be estimated analytically. Once all readings are recorded for 360 nm increase the wavelength to 380 nm.
Record the results in Data Table 2. Fluorescence and Absorbance Spectrometer Duetta is a spectrofluorometer that combines simultaneously the functions of fluorescence and absorbance spectrometers. A good overlap where peak shape and retention or migration time match indicate a pure peak while a poor overlap indicates an impure peak as demonstrated in figure 1.
This method is used in situations where sample matrix also contributes to the analytical signal a situation known as the matrix effect thus making it impossible to compare the analytical signal between sample. Repeat this procedure for all pigments in Data Table 1. It is commonly observed that differentiation degrades signal-to-noise ratio unless the.
This deviation is a consequence of a sum rule that was derived from the KramersKronig relations at a time when the fundamental limitation of Beers law was no longer mentioned in the literature.
2
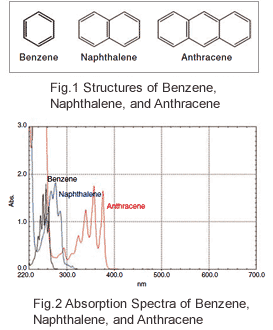
The Relationship Between Uv Vis Absorption And Structure Of Organic Compounds Shimadzu Shimadzu Corporation

What Is The Reason Of One Peak And Two Peak In An Absorbance Graph
Predicting The Redshift On The Ultraviolet Spectrum Using The Peak Area Method

Intro To Signal Processing Integration And Peak Area Measurment
Why Is It Advisable To Use The Maximal Wavelength For An Absorbance Peak When Measuring The Absorbance To Determine The Concentration Of A Compound Quora
1

Multiple Peaks In Uv Vis Absorbance Spectra What Do The Mean
