There are some clear differences between the special pairs of PSI and PSII. Since the UV absorbance also differs depend on what wavelength is used it is important to choose an appropriate wavelength based on the type of analyte.

Difference Between An Absorbance Emission And Excitation Spectrum For Uv Visible Spectroscopy Chemistry Stack Exchange
1
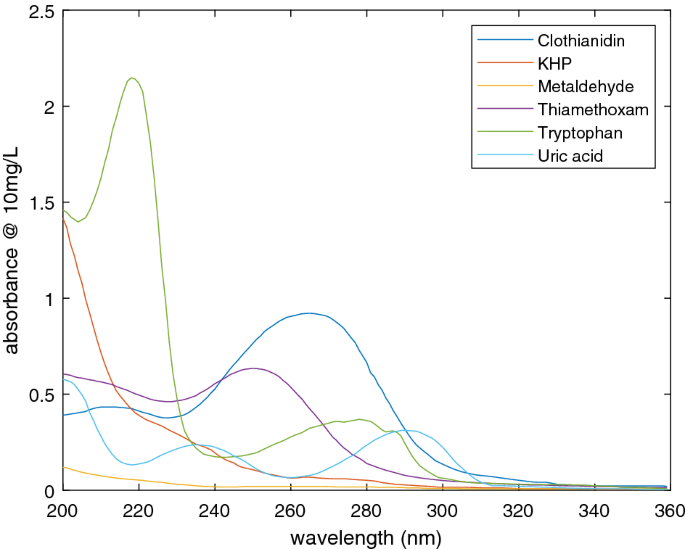
Ultraviolet Absorption Of Contaminants In Water Scientific Reports
Line is the relationship between concentration and Absorbance.

Relationship between wavelength and absorbance spectrum. Mathematically the resultant spectrum of a pulsed source can be described by a convolution between the spectrum of the light source and the spectrum corresponding to the pulses. Beers law says that the relationship between the absorbance of the chromophore and its concentration is linear allowing construction of a standard curve by plotting absorbance versus concentration such as shown in Figure 1. The optimal wavelength Amax for measuring absorbance is that wavelength that is most absorbed by the compound in question.
Figure 1 shows the relationship between the wavelength of light and the common types of electromagnetic radiation. Indeed the entire vertical absorbance scale may be changed to a molar absorptivity scale once this information about the sample is in. The relationship between HOMO-LUMO gap and absorbed wavelength of light.
The higher the value the more of a particular wavelength is being absorbed. An absorbance spectrum a plot of absorbance as a function of wavelength is determined to select the optimal wavelength for analyzing a given compound. As a result the impact of these artifacts will vary with the pulse repetition rate and the modulation depth of the light source as well as the OPD sample rate cms of the OSA.
Once the analytical wavelength for a particular solution is determined we can learn more about the solution through the relationship between absorbance A. For this process to occur the energy of the photon has to match the energy of the HOMO-LUMO gap. The general Beer-Lambert law is usually written as.
The wavelength of light where the absorbance is highest is used as the analytical wavelength. Absorbance on the vertical axis is just a measure of the amount of light absorbed. B A spectrophotometer works by splitting white light from a source into a spectrum.
Light in the short wavelengths of 200 to 400 nm is referred to as ultraviolet UV. A peak of the absorbance curve a wavelength where the absorbance reaches a maximum is where the rate of change in absorbance with wavelength is smallest. Most commonly used is 254 nm.
Lastly C is the molar concentration of the chromophore molL used for the measurement. Light can be categorized according to its wavelength. Light and Spectrum profile.
Measurements are usually made at a peak to minimize errors produced by errors in wavelength in the instrument that is errors due to having a different extinction coefficient than assumed. This provides maximum sensitivity for your measurements. I the absorption spectrum is red shifted in PSI 238-240 showing a bleaching upon oxidation at 700 nm P 700 compared to that of P 680 685 in intact systems 241-243.
A a b c where A is the measured absorbance a is a wavelength-dependent absorptivity coefficient b is the path length and c is the analyte concentration. Once you have measured E you can find the concentration of any solution of that compound by measuring Absorbance at the same wavelength used to calculate E. Relationship between the concentration of the compound being studied and its absorbance.
The optical density turbidity of the sample will depend on the wavelength so once one wavelength is chosen it must be used consistently. UV-Vis radiation has sufficient energy to cause transitions in bonding electrons as opposed to atomic innershell or valence electrons and thus is correlated best with the behavior of bonds and functional groups in the analyte. The spectrophotometer allows choice of the wavelength of light to use for the measurement.
If the isoprene spectrum on the right was obtained from a dilute hexane solution c 4 10-5 moles per liter in a 1 cm sample cuvette a simple calculation using the above formula indicates a molar absorptivity of 20000 at the maximum absorption wavelength. The Beer-Lambert law or Beers law is the linear relationship between absorbance and concentration of an absorbing species. This is the E value EAC if L1 so the slope of the standard curve gives you E.
Ii when comparing the P P or 3 PP difference spectra a very resolved spectral structure is observed in the case of P 700. You will see that absorption peaks at a value of 217 nm. This wavelength is known as λ max and is most sensitive to the changes in concentration.
A standard UV detector allows user to choose wavelength between 195 to 370 nm. This relationship is best determined using a wavelength of light in a region of the visible spectrum where the maximum absorbance is observed. The diagram below shows a simple UV-visible absorption spectrum for buta-13-diene - a molecule we will talk more about later.
Compared to a UV detector a VIS detector uses longer wavelength 400 to 700 nm. Δ A displaystyle Delta A is a function of wavelength so for a measurement to be meaningful the wavelength at which it was performed must be known. UV-Vis spectroscopy is based on the selective absorption of electromagnetic radiation in the 180-780 nm wavelength range.
The smaller the energy difference between HOMO and LUMO the less energy is needed and the longer the wavelength that will be absorbed Figure 1-1. Where Delta Absorbance is the difference between absorbance of left circularly polarized LCP and right circularly polarized RCP light this is what is usually measured.

Action Spectrum Wikipedia
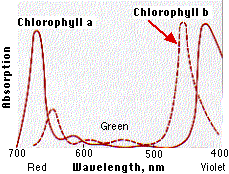
Action Absorption Spectra
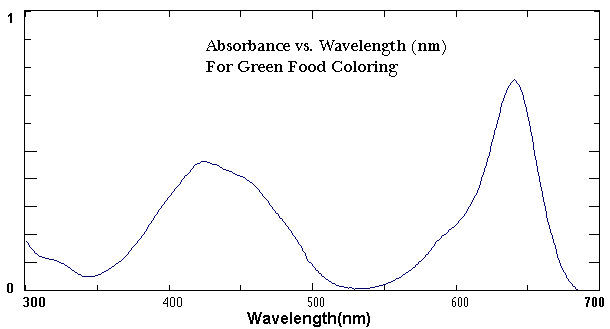
Primarylect
Chem 125 Experiment Ii
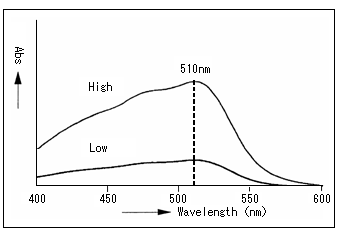
5 Colorimetric Analysis 4 Hitachi High Tech Global

The Relationship Between Absorbance And Wavelength Of Nanocomposities Download Scientific Diagram
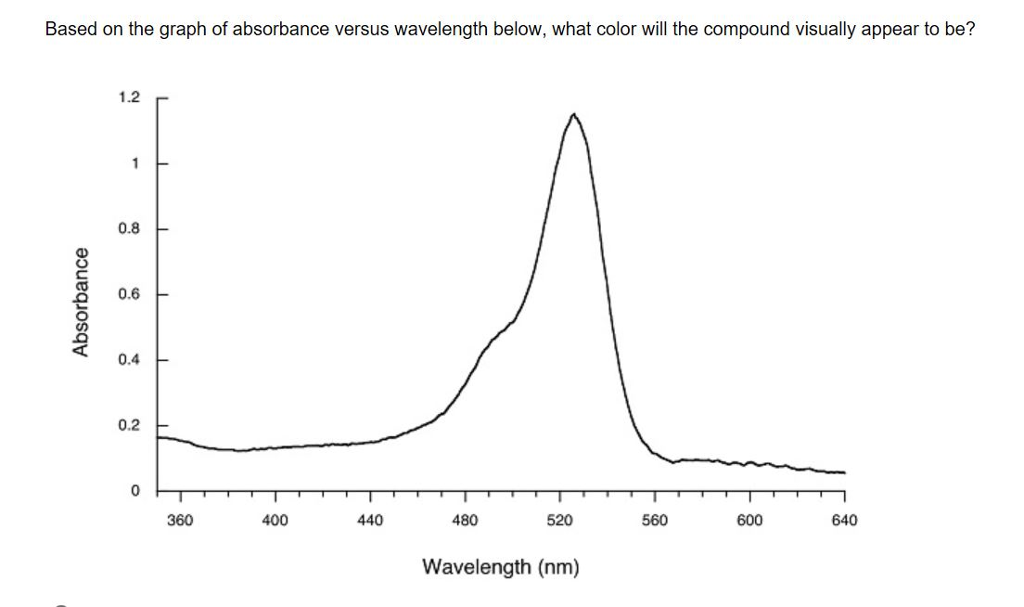
Solved Based On The Graph Of Absorbance Versus Wavelength Chegg Com
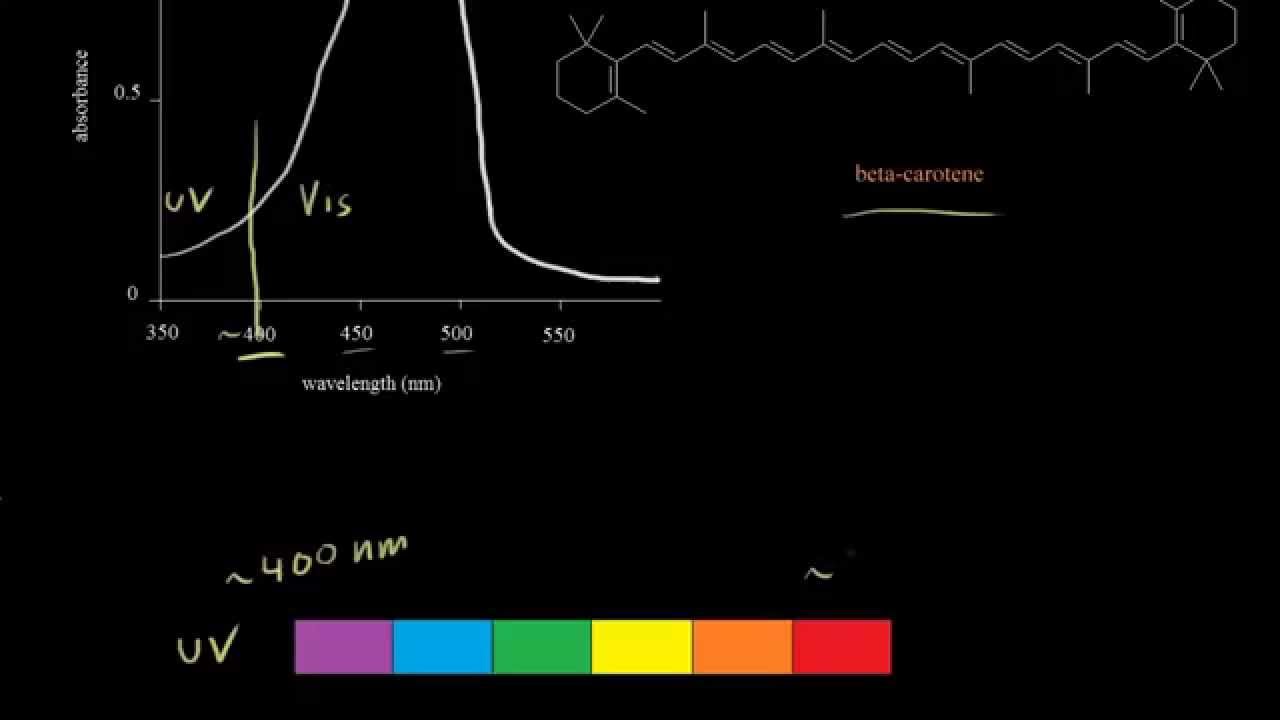
Absorption In The Visible Region Video Khan Academy
